TREAT Asia Launches a New Hepatitis C Education Campaign and Screening and Treatment Study
In January, TREAT Asia launched a new clinical study investigating how to better diagnose and treat hepatitis C in individuals co-infected with HIV in resource-limited settings. The aims of the study are to assess the need for immediate hepatitis C co-infection treatment and to develop a pilot model for treatment provision and education that can be replicated across the Asia-Pacific. “As rates of hepatitis C and HIV co-infection continue to rise around the world, managing co-infection is a critical and emerging clinical challenge,” said amfAR CEO Kevin Robert Frost
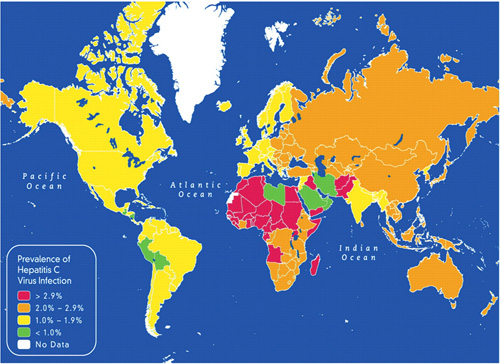
About 5 million people living with HIV, or 15% of the total HIV-positive population, are co-infected with hepatitis C, and Southeast Asia and Africa have larger numbers of co-infected individuals than other regions. These patients are more likely to develop hepatitis C-related liver disease than patients who only have hepatitis C infection.
However, the most widely available hepatitis C treatment, a 48-week standard course of pegylated interferon (an injectable medicine) and ribavirin (an oral pill), often costs between $10,000 and $20,000. Due to that high cost, the flu-like side effects the treatment combination often causes, and a lack of physician experience in managing the treatment and its side effects, many people with hepatitis C in Asia remain untreated. As a result, hepatitis C co-infection has become a significant cause of death among people living with HIV in the region.
Two new hepatitis C medicines, Sovaldi™ and Olysio™, that are easier to take and administer, were approved by the U.S. Food and Drug Administration in December, but, at approximately $80,000 per course, they are much more expensive than existing treatment, and are unlikely to become available in low- and middle-income countries in the near future. “There are people who are dying of liver failure who cannot afford to wait for newer treatments,” says Annette Sohn, M.D., amfAR vice president and director of TREAT Asia.
During the study, TREAT Asia will first screen up to 400 co-infected patients to determine their treatment eligibility. The second phase of the study will include providing up to 200 co-infected patients with free pegylated interferon and ribavirin at four partner HIV treatment centers: Cipto Mangunkusumo General Hospital in Jakarta, Indonesia; the HIV-NAT/Thai Red Cross AIDS Research Center in Bangkok, Thailand; the National Hospital for Tropical Diseases in Hanoi, Vietnam; and the University of Malaya Medical Centre in Kuala Lumpur, Malaysia. TREAT Asia has also been holding hepatitis C treatment workshops in preparation for the study, and will publicly distribute a series of easy-to-understand educational materials for patients and healthcare providers that includes animated videos, booklets, and flipcharts—available in Bahasa Indonesia, Chinese, English, Malay, Thai, and Vietnamese.
TREAT Asia is also preparing to work with the World Health Organization to disseminate their upcoming hepatitis C treatment guidelines, and is collaborating with regional partners to advocate for increasing the availability of hepatitis C co-infection treatment in Asia. “We believe that this comprehensive program of disease education, treatment preparedness, and ongoing adherence support will allow us to guide patients throughout their treatment journey, and significantly enhance the chances of individual cure and program success,” says Nicolas Durier, M.D., M.P.H., TREAT Asia’s director of research and one of the study’s principal investigators.
Share This: